Pfizer FDA Briefing Meeting Document Analysis
This article has been made into a movie
The Pfizer data we are about to review is based off 4,526 clinical subjects (allegedly) with twice as many in the vax arm than the placebo in both age brackets. Here is the source data we will review: https://www.fda.gov/media/159195/download
In a combined analysis of both age groups, claimed VE is 80.4% with 3 cases in the BNT162b2 group and 7 cases in the placebo group.
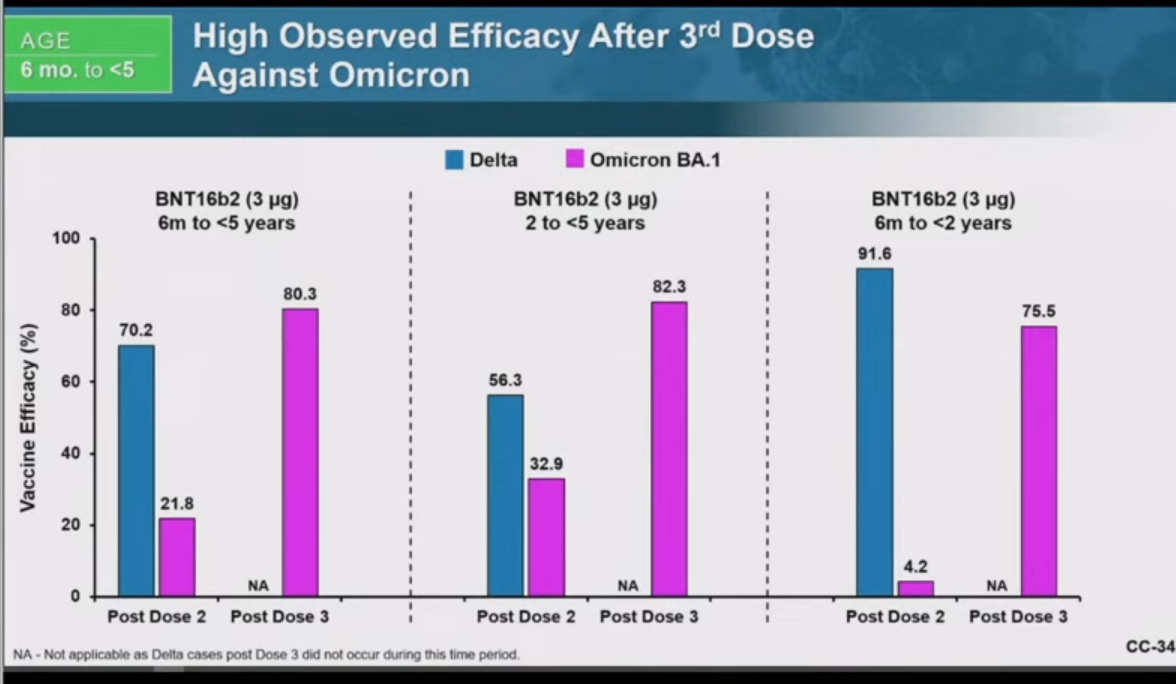
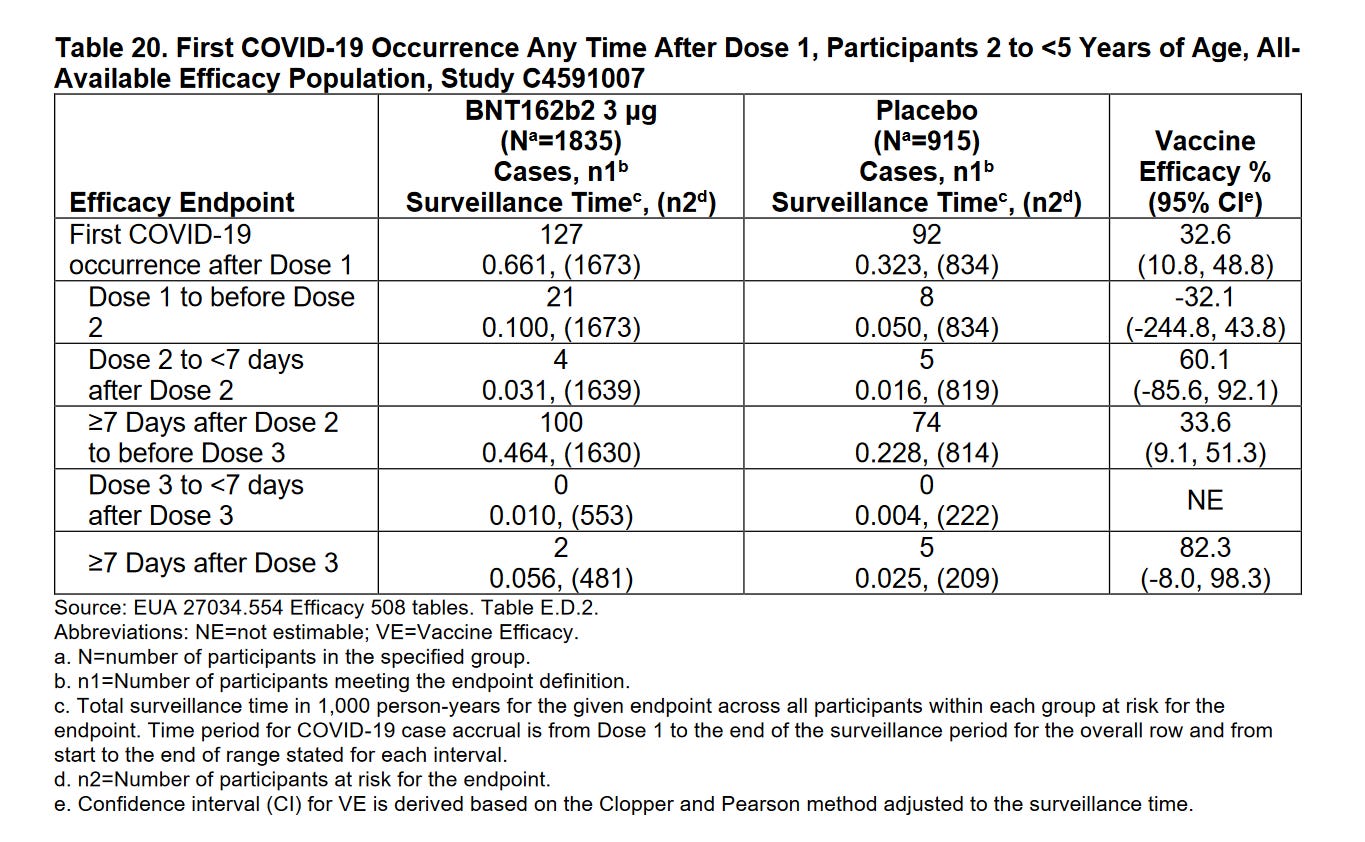
Efficacy was admittedly negative after the 1st dose: -32.1% effective in 2-5 year olds (21 cases in BNT162b2 arm & 8 cases in placebo arm) and -29.7% effective for 6-23 month olds (13 cases in BNT162b2 arm & 5 cases in placebo arm) But note how Pfizer left this off their bar charts they presented at the FDA meeting for their EUA on 6/15/2022 (which is 6/6/6 if you add the digits)
Before we get into the data for 6-23 month olds and 2-4 year olds, respectively, I wanted to remind people that the data presented was in relation to a “data cutoff” at April 29, 2022. How this date was selected is unknown, so it is very possible that this data was selected to maximize safety and efficacy.. Just like the arbitrary throwing out of all the cases before dose 2 (and the negative vax efficacy)
6-23 months of age
1,178 BNT162b2 recipients and 598 placebo recipients
The most commonly reported solicited ARs after any dose were irritability (51.2%), drowsiness (27.0%), decreased appetite (22.2%), and tenderness at the injection site (16.6%).
Local adverse reactions: 23.8% after dose 1, 21.6% after dose 2 & 20.5% after dose 3
Systemic adverse reactions: 61% after dose 1, 55.8% after dose 2 & 51.5% after dose 3
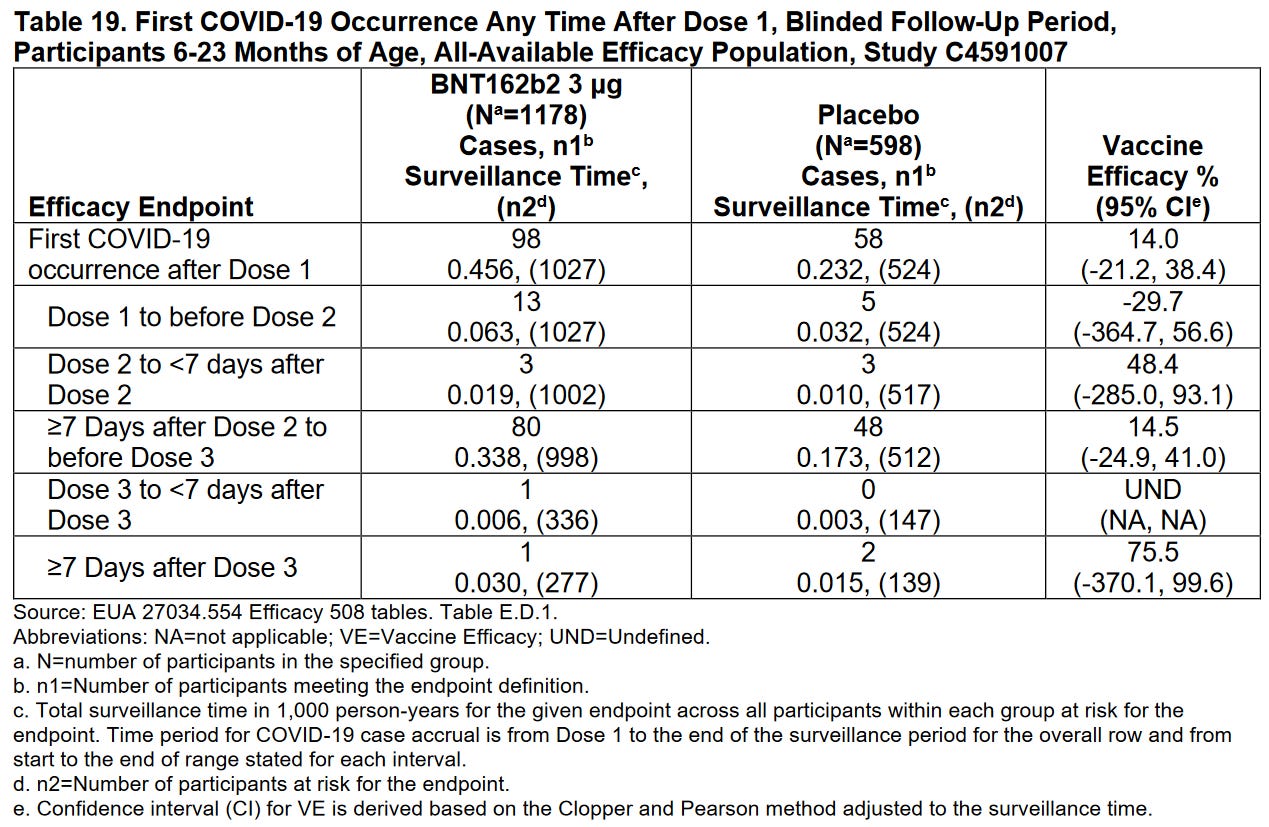
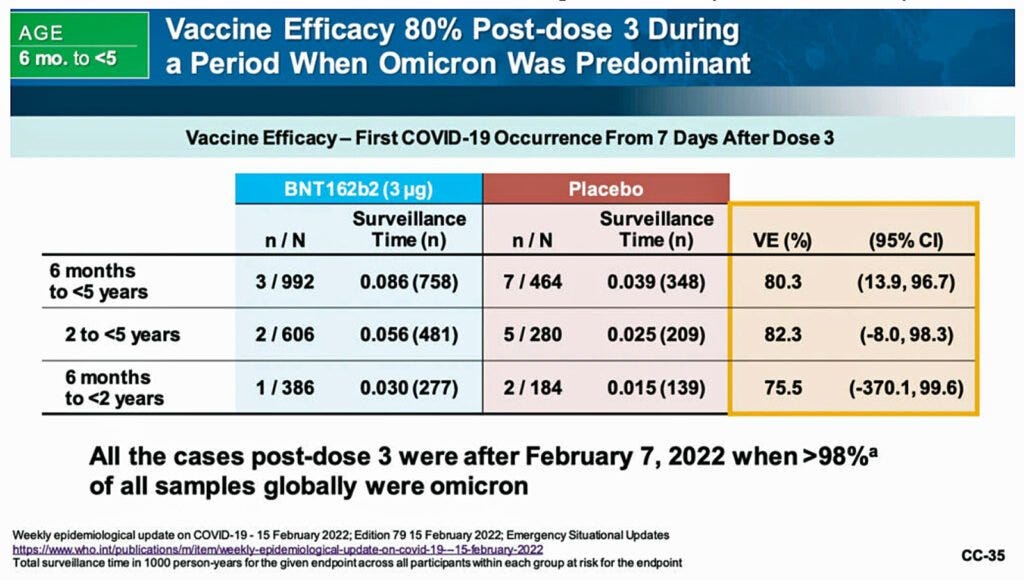
Claimed efficacy of 75.6% was derived using only 3 Covid19 cases: 1 in BNT162b2 & 2 in placebo study. Pfizer only looked at 31.25% of the subjects (376 BNT162b2 recipients and 179 placebo recipients) and counted cases of covid starting 7 days after dose 3 and ending at their April 29th cutoff date.
Six participants 6-23 months of age (3 BNT162b2 recipients and 3 placebo recipients) “developed more than one virologically and clinically confirmed episodes of symptomatic COVID-19 disease”. (unclear but possible they may have only included data from post dose 2 and before data cutoff)
From Dose 1 through the data cutoff: 1 placebo recipient 6-23 months of age “met the criteria for severe COVID-19”, 0 BNT162b2 recipients
The median duration of blinded follow-up for participants after Dose 3 was ONLY 1.3 months (range: 0-3.2 months). The median duration of combined blinded and unblinded follow-up after Dose 3 was ONLY 2.1 months
Note: 1) the totals row is deceptively first 2) UND on row 3 could also be written negative infinity though I don’t know what the 95% confidence interval would be…
Only considering cases after the 3rd dose is a sneaky way to call the above “75.6% effective”
2-4 Years of Age
1,835 BNT162b2 recipients and 915 placebo recipients
Local adverse reactions: 35.5% after dose 1, 36.3% after dose 2 & 31.5% after dose 3
Systemic adverse reactions: 38% after dose 1, 33.7% after dose 2 & 30.8% after dose 3
The most commonly reported solicited ARs after any dose were pain at the injection site (30.8%), fatigue (29.7%), and injection site redness (11.4%).
Claimed efficacy of 82.4% was derived using only 7 Covid19 cases: 2 in BNT162b2 arm & 5 in placebo arm. Pfizer only looked at 31.2% of the subjects (589 BNT162b2 recipients and 271 placebo recipients) and counted cases of Covid19 and starting 7 days after dose 3 and ending at their April 29th cutoff date.
6 participants (5 BNT162b2 recipients and 1 placebo recipient) developed more than one virologically and clinically confirmed episode of symptomatic COVID-19 disease.
6 BNT162b2 recipients “met the criteria for severe COVID-19”, and just 1 placebo recipient. “All cases occurred post Dose 2 (range 32-208 days post Dose 2), and none occurred post-Dose 3” -200% vaccine efficacy against severe COVID-19? Prof Martin Neil on Twitter: “mean efficacy is -177% [95% CI (-1059%, 58.5%)] Conclusion: The treatment causes severe disease in kids.“ I’m not sure I agree, other than, this is the same logic used to say the vaccines prevent disease in other instances
NEGATIVE INFINITY efficacy against Covid with hospitalization: One subject had to be hospitalized with a severe COVID-19 case in the BNT162b2 recipients, 99 days post-Dose 2 in a 2-year-old participant who had increased respiratory rate (RR), decreased SpO2 as severe case criteria and was hospitalized due to COVID-19. The participant reported fever, new or increased cough, and new or increased shortness of breath, with at least 1 symptom ongoing as of the last report. During the urgent care visit, the participant had hypoxemia and was hospitalized with wheeze on lung auscultation. BioFire testing was positive for parainfluenza virus 3, in addition to the positive central laboratory COVID-19 result. The participant received oxygen via nasal canula, inhaled salbutamol and oral steroids while hospitalized, then was discharged home 3 days later. In the placebo group, a 2-year-old participant met severe criteria because of decreased SpO 2 (88% on room air) with symptoms of new or increased cough and nasal congestion.
The median duration of blinded follow-up for participants after Dose 3 was ONLY 1.4 months (range: 1-3.2 months) The median duration of combined blinded and unblinded follow-up after Dose 3 was ONLY 2.1 months
“Uncertainties”
As always, pay close attention to the “unknowns” and “uncertainties” sections of these documents, usually buried off near the end of the reports.
Unknown duration of vaccine effectiveness, due to “limited” evaluation period and “waning of protection” in older groups
Unknown if booster will be needed… but “likely that a booster dose will be needed in addition to the three-dose primary series”
And if a new variant pops up, all bets are off
Unknown effectiveness against asymptomatic infection & transmission
Immunobridging:
“Immunobridging success criteria were met for both age groups of 6-23 months and 2-4 years“
Immunobridging is their future of vaccine approvals Rushed through with only minimal test tube or mice testing
All Those Extrapolations… Just 10 Cases From 7 Days After Vax #3:
Pfizer Confesses To FDA They Don’t Understand How Vax Works
Bombshell Admission! Pfizer Confesses To FDA: They DON'T Really Know How The Vax Works:
"Obviously we don't have a complete understanding of the nature of the way that the vaccine works in terms of producing immune response"